Efficiency against Salmonella, Klebsiella and MRSA - Test result Korea Conformity Laboratories
Wellisair: Efficiency against Salmonella & Klebsiella & MRSA
Bacteria test report (nº 027129): March 24th, 2016 Korea Conformity Laboratories (KCL).
Scope: measure the effectiveness of Wellisair for the surface disinfection against Salmonella Typhimurium (bacteria causing diarrhoea, abdominal pain, vomiting and nausea, which can be fatal), against Klebsiella pneumoniae (bacteria causing urinary tract infections, pneumonia, sepsis, soft tissue infections, and surgical wound infections) and against MRSA (bacteria contracted in hospitals through the insertion of a ventilator tube in the patient that produces a nosocomial pneumonia, a disease that can be fatal).
Procedure:
- Test Method: measure the reduction rate of different bacteria for 4 hours with a distance between the medium inoculated with the strain and the sample of 5cm.
- Test Conditions: 37ºC 33,1% R.H.
Results:
Salmonella Typhimurium | Klebsiella pneumoniae | MRSA | |||
Before Wellisair operating | After Wellisair operating | Before Wellisair operating | After Wellisair operating | Before Wellisair operating | After Wellisair operating |
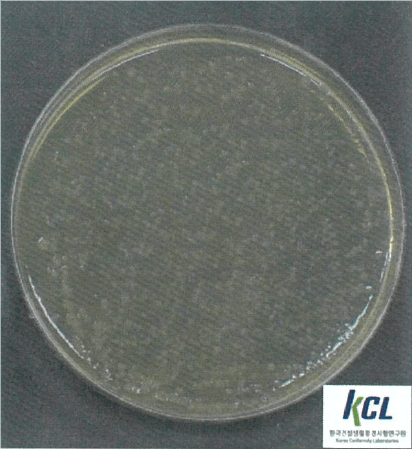 | 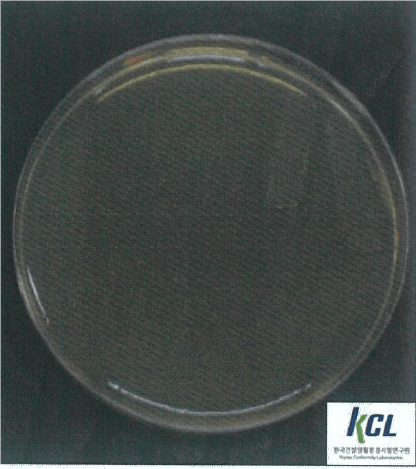 | 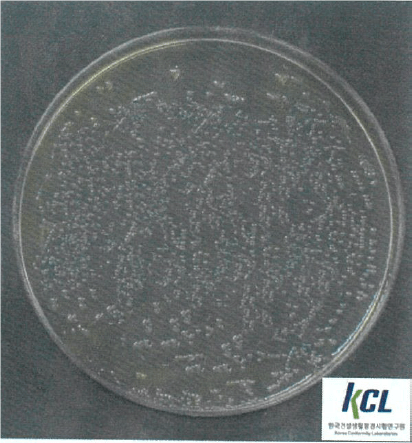 |  |  | 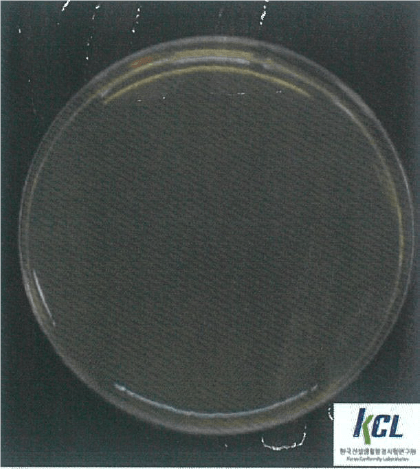 |
Test Items | Test Results | |||
Early (CFU/mL) | After 4 h (CFU/mL) | Reduction rate (%) | ||
Salmonella Typhimurium | No treatment | 1,6x104 | 1,6x104 | - |
Wellisair treatment | 1,6x104 | <10 | 99,9% | |
Klebsiella pneumoniae | No treatment | 2,0x104 | 2,0x104 | - |
Wellisair treatment | 2,0x104 | <10 | 99,9% | |
MRSA | No treatment | 1,2x104 | 1,2x104 | - |
Wellisair treatment | 1,2x104 | <10 | 99,9% | |
Conclusions: Wellisair air disinfection was able to reduce 99,9% of the initial surface concentration of Salmonella, Klebsiella and MRSA microbes after 4 hours of treatment.
